CORONAVIRUS UPDATE : for patients and families
Frequently Asked Questions about the COVID-19 Vaccine.
(also referred to as SARS-CoV-2) vaccine and treatment.
Scientists from across the world have been working since the pandemic started to make a COVID-19 vaccine available as soon as possible. There are at least 36 vaccines under investigation and 2 have been approved so far under an Emergency Use Authorization (sometimes referred to as an, EUA).
We asked our in-house experts to address some of the questions continually being asked by our patients, family members, and staff.
-
WHICH VACCINE SHOULD I GET & WHEN?
Your ARBDA Care Team is continually evaluating how the most current research will impact the lives of patients entrusted to our care. Unfortunately, the topic of vaccines (in general) is far more complicated for patients with rheumatologic diseases than the average patient. This is mainly because specific conditions and treatments can change the guidance as to when (and in whom) vaccination is appropriate.
-

WILL IT BE MANDATORY FOR EVERYONE TO RECEIVE A VACCINE?
No, the vaccine will not be mandatory. We recognize and respect that some of our patients will want more time to consider their options. We simply want everyone to be safe and provide the information to make an informed decision.
-
WHAT IS AN mRNA VACCINE?
Both the Pfizer & Moderna vaccines are messenger RNA vaccines – mRNA for short. In essence, the vaccine teaches our immune system to produce antibodies that would protect against future infection from COVID-19 without exposing us to the virus itself. However, contrary to what has been reported, the mRNA vaccines are by no means considered new.
-

HOW EFFECTIVE ARE PFIZER / MODERNA VACCINES?
- They appear to be very effective based on the information that is publicly available today [Jan. 11, 2021]. Pfizer’s study enrolled more than 43,000 individuals and reported 95% efficacy at 2 weeks after receiving the second vaccine dose. Similarly, Moderna reported that, in their study of >30,000 patients, an efficacy rate of 94.1% was demonstrated at 2 weeks after the second vaccine dose was given. . While numerically slightly different, this variation is almost identical. Might mention that the 94/95% efficacy was 1-2 weeks after the second vaccination. But efficacy may be > 50% 2 weeks after the first injection.
- Notably, they both appeared to prevent the forms of clinical illness of COVID-19 infection characterized as “serious” compared to placebo. Also important: both companies reported that the vaccine’s efficacy was consistent across age, race and ethnicity, and gender demographics.
-

HOW MANY INJECTIONS OF THE VACCINE WILL SOMEONE REQUIRE?
Both the Pfizer and Moderna vaccines require 2 (two) injections. For the Pfizer vaccine, there will be a 21-day space between the 2 vaccine doses. For Moderna, it is 28 days. Future vaccines that are currently in development may only require a single injection.
-

WHO HAS THE AUTHORITY TO APPROVE VACCINES FOR DISTRIBUTION?
- WHO HAS THE AUTHORITY TO APPROVE VACCINES FOR DISTRIBUTION?
-
BOTTOM LINE: SHOULD I GET VACCINATED?
Vaccination appears to protect against the severe symptoms from COVID-19 infection in more than 90 percent of study participants. These studies included more than 70,000 participants, respectively. Furthermore, those who have an underlying illness, such as diabetes, obesity, or chronic kidney disease – who are at higher risk to advance to more severe COVID illness – show equal protection as well. In the interest of transparency -there was one presentation of a severe infection case that developed in the Pfizer trial but none in the Moderna trial.
-

CONSIDER THE FOLLOWING QUESTIONS REGARDING THE VACCINES:
- What is my potential exposure to COVID-19?
- What would be the impact of having COVID-19 on my family, friends, or co-workers?
- What is the likelihood of the infection becoming severe?
- How have I responded to other types of vaccines in the past?
Balance these questions with what is known to be the downside of getting the vaccine.
Bear in mind: Infection is pervasive in the community, so exposure potential is getting increasingly worse.
On Jan 10th, the State of New Jersey reported > 6,000 new COVID cases in one day! More than 600 of those cases require critical care, and, sadly, 54 people died within a 24-hour window because of COVID infections.
-

HOW ABOUT SAFETY?
The mRNA vaccines from both Pfizer and Moderna DO NOT utilize live versions of the virus to immunize you. Bottom Line – Yes, some reactions to the vaccine injection can occur –including pain at the site, fatigue, muscle aches, headaches, chills, and fever –but these symptoms generally resolve within a few days. Some would say these undesired reactions actually indicate the response of your immune system. To this point, no significant safety events or reactions have been reported to the public in association with either vaccine candidate. As of now, the scientists and manufacturers do not foresee any risk that is greater than the risk of being infected with COVID-19. That said, however, with every day that passes, we get more information on these vaccines.
Hospitalized Covid-19 patients by day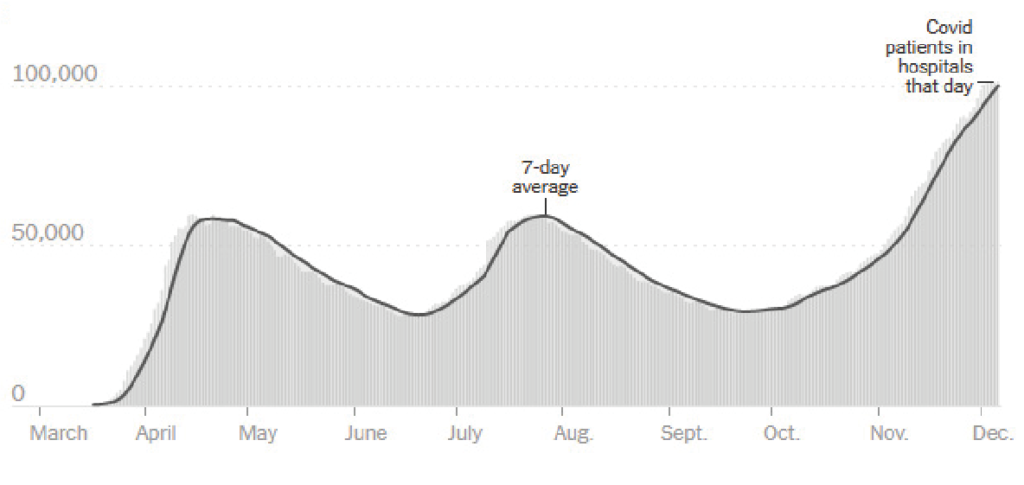
*Source: Hospitalization data from the Covid Tracking Projects. Currently hospitalized is the number of patients with Covid-19 reported by states to be in a hospital on that day. Dips and spikes could be due to inconsistent reporting by hospitals.
-

HOW CAN I FIND MORE INFORMATION ABOUT THE VACCINE?
Click on the following links from the Wall Street Journal to learn more about the individual vaccines.
▶ Wall Street Journal Summaries for Moderna and Pfizer vaccines
The Centers for Disease Control and Prevention also prepared fact sheets on vaccine safety and related topics.
Each of them is available in a print-friendly format and in five different languages, including Spanish. To change
the language, click the tab toward the top of each web page.Benefits of Getting a COVID-19 Vaccine
8 Things to Know about Vaccination Planning
Understanding How COVID-19 Vaccines Work
-

DO I STILL NEED TO WEAR A MASK AFTER VACCINATED?
Yes. While experts learn more about the protection that COVID-19 vaccines provide under real-life conditions, it will be necessary for everyone to continue using all the tools available to us to help stop this pandemic, like covering your mouth and nose with a mask, washing hands often, and staying at least 6 feet away from others.
-

WHEN CAN I STOP WEARING A MASK / SOCIAL DISTANCING FROM OTHERS?
There is not enough information currently available to say if or when CDC will stop recommending that people wear masks and avoid close contact with others to help prevent the spread of the virus that causes COVID-19.Experts need to understand more about the protection that COVID-19 vaccines provide before making that decision. Other factors, including how many people get vaccinated and how the virus is spreading in communities, will also affect this decision
-

IF ALREADY HAD COVID-19 AND RECOVERED, DO I NEED THE VACCINE?
COVID-19 vaccination should be offered to you regardless of whether you already had COVID-19 infection. You should not be required to have an antibody test before you are vaccinated. Additionally, current evidence suggests that reinfection with the virus that causes COVID-19 is uncommon in the 90 days after the initial infection. Therefore, people with a recent infection may delay vaccination until the end of that 90-day period has elapsed.
-

ADDITIONAL INFORMATION
Information regarding the COVID-19 vaccines is continuously evolving.This is the reason why ARBDA believes that discussing your vaccination options is vital to ensuring the best interests of both public health and every one of our patients.
CLICK TO VIEW ADDITIONAL INFORMATION

GUIDANCE FOR PRESCRIBED MEDICATIONS
** NOTE ** Please consult with your ARBDA physician PRIOR to stopping any treatment. The following guidance generally applies for patients currently taking: